Battery issue
i usually do not reply to moot things but i will make an exception. at present, taking care of more pressing issues. also, insulator is a generality also, but if you prefer a diff term, barrier is another one. i WILL not get into the many diff constructions because they ALL diff.
as soon as i can, will post my reply.

2. ISOLATOR:differs from a switch in that it is intended to be open when the circuit is not carrying current. FEEL FREE TO USE THE DICTIONARY. 3. SURFACE CHARGE:lead acid batteries are slow and cannot convert lead to lead dioxide quickly during charge. this delay cause the charge to lay on top of the plates which causes a false rise in SoC (state of charge). to normalize the SoC, a drain is placed across the battery for a few seconds. i always do TWO back to back load test, one to remove the surface charge and the next for battery condition.
4. STRATIFICATION:the electrolyte of a stratified battery concentrates at the bottom and starves the upper half of the cell. stratification occurs when the battery is kept below 80% and never receives a full charge. making short trips with heavy power drain adds to the issue as the alternator cannot supply a saturated charge level.light acid on top limits plate activation, promotes corrosion and reduces performance. high concentration on the bottom makes the battery appear more charged than it is and artificially raises open circuit voltage.the differences causes CCA reduction and starting is sluggish.
5. SPECIFIC GRAVITY:key parameter for battery operation. it is the ratio of weight of a solution to that of water in equal amounts at a set temperature. in this case, sulfuric acid and water. specific gravity is an indicator of SoC BUT NOT the battery's capacity. during discharge the SG decreases linearly. NOT GETTING INTO MATH SG decreases with discharge to a value nearer to water (1) and increase with charging. the electrolyte level also effects SG. low levels increase SG and adding water decreases SG. temperature also has its effects. SG is chosen by the MFG as determined by application. it is normally around 1.285 but can go higher to increase amp/hour capacity, aka, 1.320. the battery is still 62.48% water fully charged. for fools that do not do proper maintenance, a 1.6 SG acid can be used to overcome a diluted electrolyte by over filling but highly recommended only to be done by TRAINED personnel! higher SG =
1. more capacity
2. shorter life3. higher momentary discharge rates4. less adaptable to float operation 5. more standing loss
lower SG=
1. less capacity
2. longer life3. lower momentary discharge rates4. more adaptable to floating operation 5. less standing loss
THE BATTERY IS CONSIDERED FULLY CHARGED WHEN THE SG REACHES ITS HIGHEST POSSIBLE VALUE. once this is achieved, the battery becomes balanced, the electrolyte becomes a barrier or isolator to ion exchange.now you can force current through the electrolyte but when you do, the resistance of the cell turns the energy into heat which if unchecked, will destroy the cell/battery. the battery cannot charge any higher since there is no material left to exchange ions (chemical reaction between plate material and electrolyte).as i said in the beginning, EMF is a powerful force and will slowly cause a reverse of ion exchange in a properly stored and charged battery. as time goes on, it snowballs and the battery chemistry reversed itself at a rate of 1>5% monthly (variables have effects). i still have some things to expound upon but would make it difficult reading so i will post another time.
VOLTAGE METHOD: it is simple and practically everyone has a meter to do so. however it is inaccurate as hell! why?? cell material and temperature (ex: the addition of calcium which makes batteries maintenance free raises voltage levels 5>8%).the most blatant error is taking the reading when the battery is disturbed by charging or discharging as it distorts the voltage reading. to be ACCURATE, battery needs to be in a steady state rest for at least 4 hours and some mfg's suggest 24 hours for the lead acid types. this makes voltage reading SoC impractical for an active battery. also, different battery types have their own parameters so one size does not fit all! also temperature effects voltage, heat raises it and cold lowers it. also surface charge, stratification and electrolyte/plate composition and impurities. if you measure SoC via voltage, the battery must be floating with no load attached. any parasitic loads puts the battery in a quasi-closed circuit voltage condition.
HYDROMETER METHOD: obviously the battery has to have access plugs. when a lead acid battery accepts a charge, the sulfuric acid gets heavier, causing the SG to increase. when the battery discharges, the SoC decreases as the sulfuric acid removes itself from the electrolyte and binds to the plate, forming lead sulfate. the density of the electrolyte becomes lighter and more water like and the SG gets lower. the BCI (battery council international) specifies a fully charged battery at 1.265 SG but mfg's may go for 1.280 and higher. a higher SG will improve battery performance but may shorten it life due to increased corrosion.a low fluid level will change SG as water evaporates raising SG because of higher concentration. if the battery is over filled it will lower SG. when adding water, time must pass for it to be well mixedSG specifications will vary with type and application of the battery.NOTHING IN THE BATTERY WORLD IS ABSOLUTE!stratification, surface charge condition have their own effects.
COULOMB COUNTING: the meter uses ampere/second method by counting in/out current. this is an elegant solution but has flaws. for ONE, current IN will never be current OUT because there are inherit energy losses. the system does work well but the meter must be calibrated. some meters are self learning as it will use previous discharge rate to new rate and can compensate for aging and other variables. some can use new charge rate/time because a faded battery charges more quickly than a good one.
CAT'S MEOW!!!
IMPEDANCE SPECTROSCOPY: this system not only measures SoC but CAPACITY as well. it uses an AC current at specified frequencies and pretty much ignores the faults of other methods. since it is an ac current, it can measure impedance both ways in the battery. a battery is a near perfect power source as it internal resistance is almost nill and can provide massive current over time. the meter measures the impedance and as the battery ages, the impedance changes. the higher the impedance, the worst the performance gets.
Last edited by bustert; May 31, 2018 at 03:55 PM.
1. CATALYST: a. substance that enables a chemical reaction to proceed at a faster rateb. an agent that provokes or speeds significant change or reaction ex: he was a good catalyst to a lasting friendship.
2. ISOLATOR:differs from a switch in that it is intended to be open when the circuit is not carrying current. FEEL FREE TO USE THE DICTIONARY.
3. SURFACE CHARGE:lead acid batteries are slow and cannot convert lead to lead dioxide quickly during charge. this delay cause the charge to lay on top of the plates which causes a false rise in SoC (state of charge). to normalize the SoC, a drain is placed across the battery for a few seconds. i always do TWO back to back load test, one to remove the surface charge and the next for battery condition.
4. STRATIFICATION:the electrolyte of a stratified battery concentrates at the bottom and starves the upper half of the cell. stratification occurs when the battery is kept below 80% and never receives a full charge. making short trips with heavy power drain adds to the issue as the alternator cannot supply a saturated charge level.light acid on top limits plate activation, promotes corrosion and reduces performance. high concentration on the bottom makes the battery appear more charged than it is and artificially raises open circuit voltage.the differences causes CCA reduction and starting is sluggish.
5. SPECIFIC GRAVITY:key parameter for battery operation. it is the ratio of weight of a solution to that of water in equal amounts at a set temperature. in this case, sulfuric acid and water. specific gravity is an indicator of SoC BUT NOT the battery's capacity. during discharge the SG decreases linearly. NOT GETTING INTO MATH SG decreases with discharge to a value nearer to water (1) and increase with charging. the electrolyte level also effects SG. low levels increase SG and adding water decreases SG. temperature also has its effects. SG is chosen by the MFG as determined by application. it is normally around 1.285 but can go higher to increase amp/hour capacity, aka, 1.320. the battery is still 62.48% water fully charged. for fools that do not do proper maintenance, a 1.6 SG acid can be used to overcome a diluted electrolyte by over filling but highly recommended only to be done by TRAINED personnel! higher SG =
1. more capacity
2. shorter life3. higher momentary discharge rates4. less adaptable to float operation 5. more standing loss
lower SG=
1. less capacity
2. longer life3. lower momentary discharge rates4. more adaptable to floating operation 5. less standing loss
THE BATTERY IS CONSIDERED FULLY CHARGED WHEN THE SG REACHES ITS HIGHEST POSSIBLE VALUE. once this is achieved, the battery becomes balanced, the electrolyte becomes a barrier or isolator to ion exchange.now you can force current through the electrolyte but when you do, the resistance of the cell turns the energy into heat which if unchecked, will destroy the cell/battery. the battery cannot charge any higher since there is no material left to exchange ions (chemical reaction between plate material and electrolyte).as i said in the beginning, EMF is a powerful force and will slowly cause a reverse of ion exchange in a properly stored and charged battery. as time goes on, it snowballs and the battery chemistry reversed itself at a rate of 1>5% monthly (variables have effects). i still have some things to expound upon but would make it difficult reading so i will post another time.
1. is correct but fails to mention that the catalyst is unchanged.
2. An Isolator is not an insulator.. If you were to say an AGM battery uses glass mat to isolate the plates you'd be correct.
3. Charging goes not convert Pb to Pb02.. It converts PbS04 to PbO2 (Positive plate) and PbS04 to Pb (Neg plate)
The reaction.. Adding current (charging) reverses the reaction.
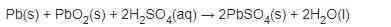
4. Good stuff.. Over 15 years I've blown $15,000 on batteries due to insufficient charge rate. Best way to reduce stratification is to equalize (over charge) the battery. The problem is that this really doesn't work on AGM batteries as they can dry out and the mat keeps the problem from occurring.
5. Specific gravity stuff is not hard to understand. H2SO4 is heavier than H20. The percentage of H2SO4 to water sets the electrolite density.
Batteries have internal leakage, At higher temps they discharge faster.
From the HD battery install sheet.
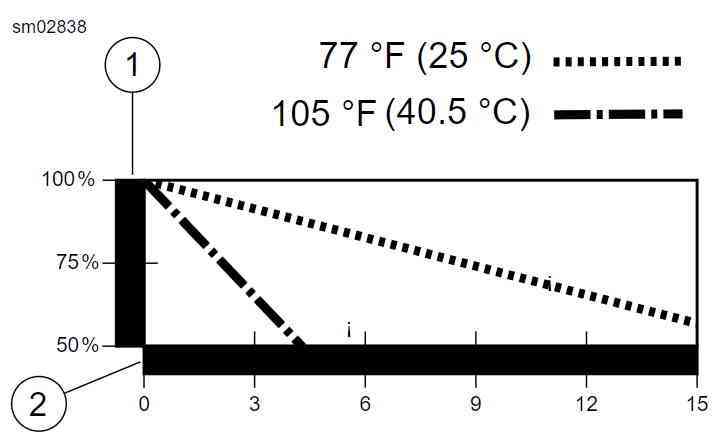
If the battery was low to start with and not charged over winter, there can be serious harm done.
you did READ i was not inclusive of all battery types, so ramble on, i consider it MOOT, you do know what that means or do i need to define!
done deal!
Last edited by tibadoe; May 31, 2018 at 04:25 PM.
The Best of Harley-Davidson for Lifelong Riders